Nukleotidy: Comprehensive Guide to Structure, Function, and Biological Significance
In the intricate world of molecular biology, nukleotidy (nucleotides) are fundamental building blocks that drive life at the cellular level. Nucleotides are organic molecules composed of a nitrogenous base, a five-carbon sugar, and one or more phosphate groups. They play crucial roles in energy transfer, cellular signaling, and the storage and expression of genetic information. Understanding nukleotidy is essential for grasping how cells function, how genetic material is replicated and expressed, and how energy is managed in biological systems.
Nukleotidy are not only structural components of nucleic acids like DNA and RNA, but they also participate in critical metabolic processes. Molecules such as ATP (adenosine triphosphate) act as cellular energy currency, while cyclic nucleotides like cAMP function as intracellular messengers in signal transduction pathways. The study of nukleotidy spans biochemistry, genetics, molecular biology, and medicine, revealing their central role in maintaining life. This article explores nukleotidy in depth, examining their structure, types, biological functions, synthesis pathways, clinical relevance, and potential applications in research and biotechnology.
Structure of Nukleotidy
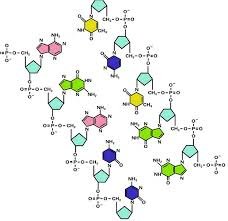
A nucleotide consists of three main components: a nitrogenous base, a pentose sugar, and a phosphate group. The nitrogenous base can be a purine (adenine or guanine) or a pyrimidine (cytosine, thymine in DNA, or uracil in RNA). The pentose sugar is either ribose in RNA or deoxyribose in DNA. Phosphate groups attach to the 5’ carbon of the sugar, forming high-energy bonds essential for energy transfer and polymerization in nucleic acids.
The combination of these components allows nucleotides to perform dual roles. In nucleic acids, the sugar-phosphate backbone provides structural integrity, while the nitrogenous bases carry genetic information through complementary base-pairing. This modular architecture also enables nucleotides to act as carriers of chemical energy, signaling molecules, and cofactors in enzymatic reactions, making them versatile molecules central to cellular physiology.
Types of Nukleotidy
Nukleotidy can be classified based on their function, nitrogenous base, or phosphate content. The primary types include:
- ATP (Adenosine Triphosphate) – The main energy currency of cells, storing and transferring energy for metabolic reactions.
- GTP (Guanosine Triphosphate) – Participates in protein synthesis, signal transduction, and microtubule dynamics.
- CTP (Cytidine Triphosphate) and UTP (Uridine Triphosphate) – Essential for RNA synthesis and lipid metabolism.
- dNTPs (Deoxyribonucleoside Triphosphates) – Serve as building blocks for DNA synthesis during replication.
- cAMP and cGMP (Cyclic Nucleotides) – Function as second messengers in intracellular signaling pathways.
Each type of nucleotide has unique biochemical roles, reflecting the diversity and specialization of these molecules in cellular processes. Their versatility underscores the importance of nukleotidy in both structural and functional aspects of biology.
Biological Functions of Nukleotidy
Nukleotidy serve a wide range of biological functions critical to life:
- Genetic Information Storage and Transfer: DNA and RNA are polymers of nucleotides that encode genetic information, enabling heredity, gene expression, and protein synthesis.
- Energy Metabolism: ATP hydrolysis provides energy for cellular activities, including muscle contraction, active transport, and biosynthesis of macromolecules.
- Signal Transduction: Nucleotides like cAMP and cGMP act as intracellular messengers, mediating responses to hormones, neurotransmitters, and environmental stimuli.
- Cofactors in Enzymatic Reactions: Nucleotides serve as precursors for essential cofactors like NAD+, FAD, and Coenzyme A, participating in oxidation-reduction reactions and metabolic pathways.
Through these functions, nukleotidy maintain cellular homeostasis, regulate physiological processes, and ensure proper communication within and between cells.
Nucleotide Synthesis: De Novo and Salvage Pathways
Cells generate nucleotides through two primary pathways: de novo synthesis and salvage pathways.
- De Novo Synthesis involves building nucleotides from small molecules such as amino acids, carbon dioxide, and ribose-5-phosphate. This process is energy-intensive but essential for rapidly dividing cells. Purine nucleotides are synthesized stepwise from inosine monophosphate (IMP), whereas pyrimidine nucleotides are assembled from orotate precursors before attachment to ribose-5-phosphate.
- Salvage Pathways recycle preformed bases and nucleosides from cellular degradation or diet, reducing energy expenditure. Enzymes like hypoxanthine-guanine phosphoribosyltransferase (HGPRT) catalyze the reattachment of bases to ribose-phosphate, conserving resources while maintaining nucleotide pools.
Together, these pathways ensure a continuous supply of nucleotides for DNA/RNA synthesis, energy metabolism, and signaling functions.
Role of Nukleotidy in DNA and RNA
Nucleotides polymerize to form DNA and RNA, with the sugar-phosphate backbone providing structural support and nitrogenous bases encoding genetic information. Complementary base-pairing (A-T/U, G-C) allows precise replication and transcription, ensuring fidelity in genetic transfer.
In DNA, deoxyribonucleotides form double helices, stabilizing the genome and allowing storage of hereditary information. RNA nucleotides, incorporating uracil instead of thymine, form single-stranded molecules that translate genetic codes into functional proteins. Beyond structural roles, nucleotides in nucleic acids are dynamic participants in regulatory mechanisms, such as RNA splicing, ribozyme activity, and epigenetic modifications.
Nucleotides in Energy Transfer and Metabolism
ATP, often called the “energy currency of the cell,” powers numerous biochemical reactions. Hydrolysis of the high-energy phosphate bonds releases energy, driving anabolic reactions, active transport, and cellular motility. GTP serves similarly in processes like protein synthesis and intracellular signaling, while other nucleotides participate as coenzymes in metabolic cycles.
The dynamic turnover of nucleotides ensures that cells can respond rapidly to energy demands, maintain metabolic flexibility, and coordinate complex biochemical networks. Imbalances in nucleotide metabolism can lead to disorders, highlighting their critical role in maintaining physiological homeostasis.
Clinical Relevance and Therapeutic Applications
Abnormalities in nucleotide metabolism are associated with various diseases, including immunodeficiencies, gout, cancer, and viral infections. For example, mutations in HGPRT cause Lesch-Nyhan syndrome, characterized by uric acid overproduction and neurological symptoms. Targeting nucleotide pathways also provides therapeutic opportunities: antiviral drugs like acyclovir and antimetabolites like 5-fluorouracil exploit nucleotide analogs to inhibit viral replication or cancer cell proliferation.
Nucleotides also have applications in biotechnology and molecular medicine. Synthetic nucleotides are used in PCR, DNA sequencing, gene editing, and as molecular probes, demonstrating their versatility beyond physiological roles.
Future Perspectives in Nucleotide Research
Research on nucleotides continues to expand, exploring novel signaling pathways, nucleotide analogs for therapeutics, and synthetic biology applications. Advances in understanding nucleotide-mediated regulation, epigenetic mechanisms, and energy metabolism could lead to innovative treatments for metabolic disorders, cancer, and infectious diseases.
Synthetic biology efforts aim to engineer artificial nucleotide analogs and polymerases, expanding the potential for novel genetic codes and bioengineering applications. As nucleotide research progresses, it promises to uncover new insights into cellular function and offer transformative tools for medicine and biotechnology.
Conclusion
Nukleotidy are central to life, functioning as the building blocks of genetic material, carriers of cellular energy, and mediators of intracellular signaling. Their diverse roles, ranging from DNA/RNA synthesis to energy transfer and signal transduction, highlight their indispensable contribution to biology.
Understanding nucleotide structure, function, synthesis, and clinical significance provides critical insights into cellular physiology, molecular genetics, and therapeutic innovation. As research advances, nukleotidy will continue to play a pivotal role in medicine, biotechnology, and fundamental science, underscoring their enduring importance in both natural and applied biological systems.
FAQs
What are nukleotidy (nucleotides)?
Nukleotidy are organic molecules composed of a nitrogenous base, a pentose sugar, and phosphate group(s) that form the building blocks of DNA and RNA and participate in energy metabolism and signaling.
What are the main types of nucleotides?
Major types include ATP, GTP, CTP, UTP, dNTPs, and cyclic nucleotides like cAMP and cGMP, each serving roles in energy transfer, nucleic acid synthesis, or signal transduction.
How are nucleotides synthesized in cells?
Cells synthesize nucleotides via de novo pathways from small precursors or salvage pathways by recycling preformed bases and nucleosides.
Why are nucleotides important for energy?
ATP and other nucleotides store chemical energy in high-energy phosphate bonds, which is released to power cellular processes such as biosynthesis, transport, and motility.
Can nucleotides be used therapeutically?
Yes, nucleotide analogs are used in antiviral therapy, cancer treatment, molecular biology, and biotechnology for research, diagnostics, and synthetic biology applications.